Augmented Reality Navigation in TKRAt Landmark Hospitals

Telugu super news,Hyderabad, april 18,2024:Total Knee Replacement (TKR) is a very common and highly successful surgery. Success rates are over 95% and average durability is over 20 – 25 years. In India every year about 3 lakh TKRs are performed.
To improve the outcomes further ‘accuracy’ of implant placement is one way. To do that several
techniques were developed.
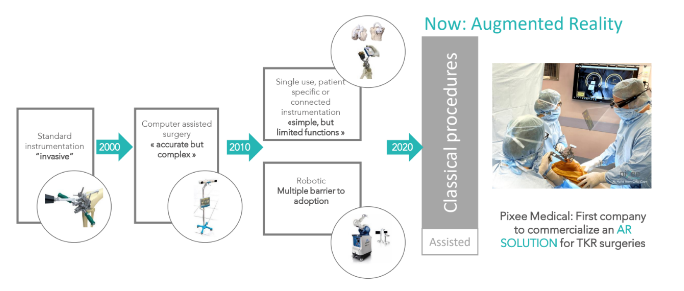
What is AUgmented Reality TKR technology:
Speaking to the media Dr. Sudhir Reddy, Chief Orthopeadic Surgeon, Landmark Hospitals
said, by using the cutting edge tracking system and Augmented Reality tools to give the
surgeon ability to achieve accurate alignment, projects Augmented Reality vision to the surgical
field, making accurate placement of implants seamless without having to use invasive tools.Also
the surgeon is in total control of the procedure white the technology only assists or guides.

AR technology s the latest and most advanced technological development in the field of TKR. It
gives the accuracy of Robotic TKR without the damage and restrictions associated with Robotic
TKR. Also because of perfect alignment in implant fixation, the longevity of TKR is likely to
increase.
AR TKR Advantages over Robotic TKR
Augmented Reality TKR Robotic TKR
Accuracy Good Good
Invasive (damage to tissues) Less More
Incision Small Large
Complications such as
frcature, infection
Less More
Surgeon Control Full Partial
Implant Choice Complete No choice
Radiation risk Nil Yes (CT scan)
In Telangana and Andhra Pradesh – 1st Augmented Reality TKR was conducted at
Landmark hospitals on 27th Feb’24 by Dr Sudhir Reddy’s team. Now nearly 70 surgeries
have been done using AR technology.

Patient experiences:
Mr Prasad Reddy, 64 yrs had bilateral TKR using AR technology one week ago. He says ‘ I
underwent both knee replacement surgeries two days ago. I have very minimal pain and already
able to walk independently without any support. I am discharged with in two days. I am able to
climb stairs also. I am surprised at such quick recovery and minimal pain.
Mrs Saroja, 58 yrs said ‘after undergoing TKR using AR technology I got Discharged With in
one day and have been walking independently. After 2 days I even did some work in kitchen.
Initially i was so afraid of pain and had postponed the surgery for a long time and suffered. Now
i feel like it is new life and I am surprised at how quick the recovery is.

























